This observation is connected to the discrete nature of the allowed energies of a quantum mechanical system. It is observed that line spectra discussed in the previous sections show that hydrogen atoms absorb and emit light at only discrete wavelengths. Bohr’s model required only one assumption: The electron moves around the nucleus in circular orbits that can have only certain allowed radii. Date: 26 August 2010, 08:58 (UTC) Source: HSCirkel.svg StylisedLithiumAtom.svg Author: HSCirkel. 
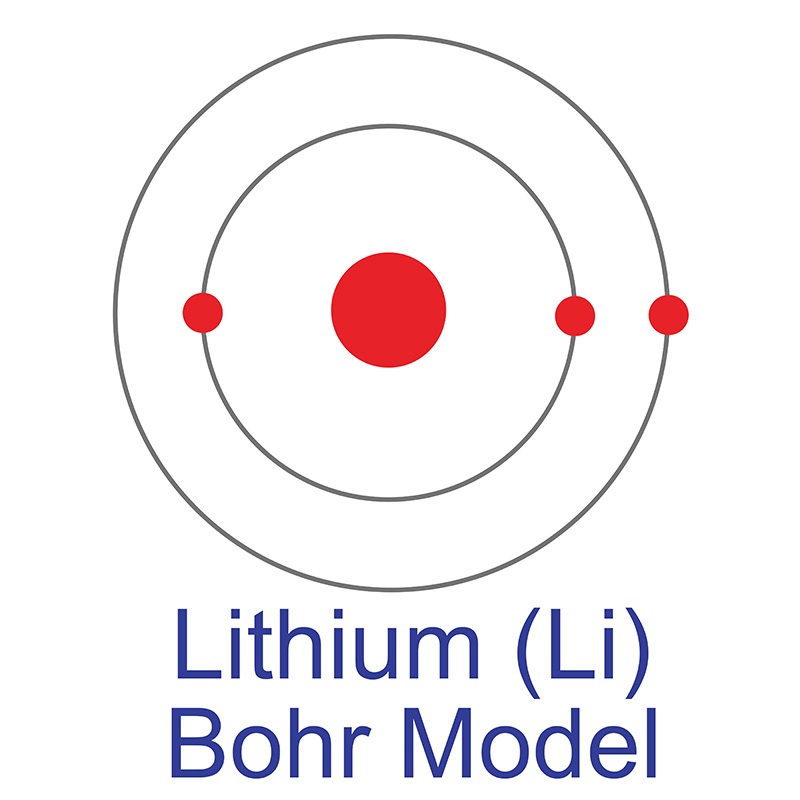
Black dots are electrons, red dots are protons and blue dots are neutrons. K, L, and M shells as 2, 8, and 6, respectively. SVG by Indolences Modification de Image:Stylised Lithium Atom.png par Liquid2003. The protons and neutrons are placed inside the nucleus while the electrons are housed in three shells i.e. Hence, the final Bohr model of the chlorine atom consists of 18 neutrons and 17 protons in the nucleus, and 17 electrons revolving in the group of 2, 8, and 7, in the K, L, and M shell, respectively. In each of these energy levels, electrons move in circular orbit. Conclusion The final Bohr model of the Sulfur atom consists of 16 protons, 16 neutrons, and 16 electrons. Each energy level has a definite energy associated with it. 
The electron in an atom has only certain definite stationary states of motion allowed to it, called as energy levels. From this, it is easy to see that like charges (charges of the same sign) repel each other. In 1913, a Danish physicist, Niels Bohr (18851962 Nobel Prize in Physics, 1922), proposed a theoretical model for the hydrogen atom that explained its emission spectrum. The Bohr model shows that the electrons in atoms are in orbits of differing energy around the nucleus. If \(Q_1\) and \(Q_2\) are the same sign, then the curve which is a purely repulsive potential, i.e., the energy increases monotonically as the charges are brought together and decreases monotonically as they are separated.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
